 Not represented by any simple chemical formula, unlike if it were a This implies that components of air are easily separable by physical methods.Ī mixture of carbon(IV) oxide, nitrogen, oxygen, water vapor and noble gases in appropriate ratio does not produce any observable change identifiable with chemical reactions (such as evolution of heat, explosion and volume change), but the mixture is similar to ordinary air in everyway. When liquid air is heated, nitrogen evaporates earlier, leaving almost pure oxygen. The dissolution and release of air from water is a physical process which implies that air is a mixture. The increase in percentage of oxygen only shows that water usually contain dissolved oxygen, even more than nitrogen (oxygen is about twice more soluble in water than nitrogen). If air is dissolved in water and boiled out again, it will be observed that the percentage of oxygen in the air is increased from 21% to about 30%. This implies that if air were a compound, its composition would be definite or constant. Variations in composition have been found when samples of air are taken from different parts of the earth. The composition of air is not quite constant. The following observations confirm air to be a mixture: The house bears witness to a new way to use industrial buildings for cultural purposes, and symbolizes a changing Odda.Air is a mixture of gases - nitrogen, oxygen, carbon(IV) oxide, water vapor and noble gases. When the production at the Odda Smelting plant was discontinued, the Linde Building was put to use for cultural events as theatre productions, concerts and the annual literature festival. The architecture reflects the styles adopted in industrial buildings in the late 19th and early 20th century, when modernism was gaining ground, but classicist elements still prevailed. The Linde Building was in a state of bad disrepair when the NVIM museum, national heritage institutions and the local municipality together made it possible to restore a large part of the façade. In the article “Harnessing the Hottest Heat and the Coldest Cold”, a story about the Odda factories from 1918, nitrogen is described as ”the World`s Friend”. The demand for nitrogen has been great in our age, because of the use of chemical fertilizers in farming, to increase the harvest. His LNG freezing technology patent has played a very important role for the international commerce of natural gas, and his research has been crucial for all cooling technology. 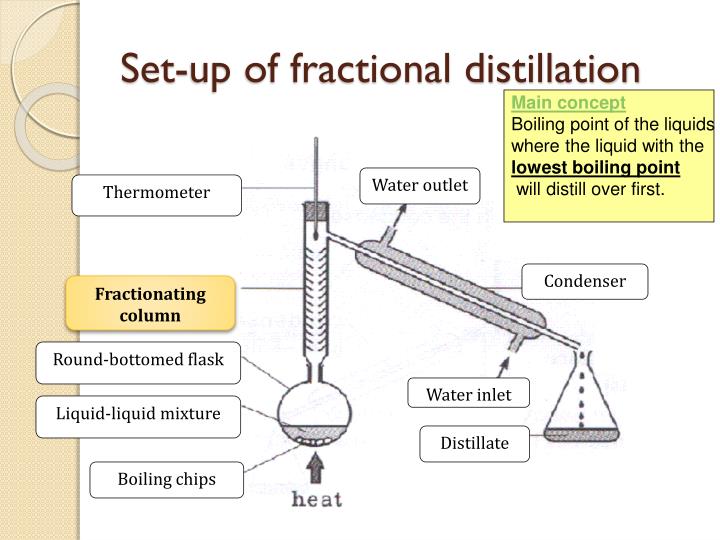
The process was discovered in 1897, when Von Linde managed to make oxygen, nitrogen and air liquid in one and the same operation. Von Linde is recognized as the inventor of the commercial LNG process which turns dry gas into a liquid by means of a combination of increased pressure and cooling. Albert Petersson, wrote his PhD thesis on the Thomson effect in Zürich in 1895. The first manager of the Smelting Plant, Dr. The Linde plant is based on the so-called Joule-Thomson effect. Not long after Hampson invented a similar method in London, independently of Linde. The most important method for producing liquid air is the one invented by Linde in Munich in 1895. The last compressors, which were replaced in the Nineties, have been removed and sold. This is the plant which remains in the building today. After World War II the plant was obsolete and unreliable, and a new nitrogen plant with new compressors was bought and installed in 1949. In 1915 the daily production was 100 tonnes liquid air, which was distilled to make 77 tonnes nitrogen. A new plant was built in 1912, when the production capacity of the smelting plant was increased. The processing plant was set up when production was started in 1907/08. It concerned the cyanamide factory and its pure nitrogen production plant. The first building permit for the Linde plant was issued on. Crushed carbide was combined with other substance in cylinder-shaped kilns, into which nitrogen from the Linde plant was conveyed through pipes in the ground below the hall housing the kilns. The Linde plant was installed right alongside the calcium cyanamide factory, because the nitrogen from the Linde Building was needed in the cyanamide kilns. The air intake was not far from the school, some distance from the factory. To produce calcium cyanamide, one must have calcium carbide and nitrogen gas. The oxygen is distilled and the nitrogen remains. At a certain point the air becomes liquid. The process is repeated, and can be used to achieve very low temperatures. 
Put in a nutshell the process consists of compressing the air and then allowing it to expand. Air consists of about 20 per cent nitrogen and 80 per cent oxygen. The Linde plant is a large cooling system where oxygen and nitrogen are separated by fractional distillation and liquid air is produced. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
